MARI on Map
Testimonial
FAQ
What is the importance of MARI organization goals, and how does the world/health care system benefit from this?
This essay highlights the important global-scale contribution that MARI plays in many
different fields, such as technology, pharmaceutical, nutrition, law, and veterinary to
name a few. However, the key emphasis of this paper is focused on the role that
MARI plays in the field of healthcare. This paper addresses how misdiagnosis is a
large but silent problem that exists within the healthcare community and how through
the resources that MARI provides help to reduce the rates of misdiagnosis. MARI
aids in decreasing the rate of misdiagnosis by providing short, effective, informative
summaries of medical conditions that educate the patients and increases the ability
of the healthcare providers to formulate an accurate diagnosis.
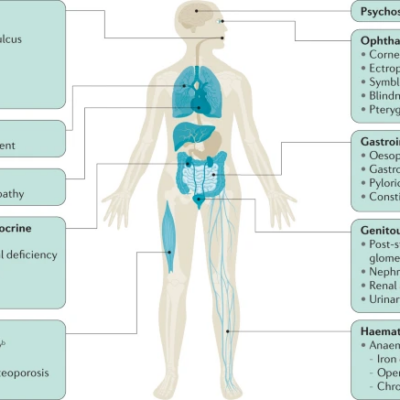
A common proxy used to measure the quality of healthcare in different countries is to
measure the outcome of treatable diseases, including diagnostic errors, which is
defined as a diagnosis that is missed or wrongly diagnosed within the healthcare
system (Martin, 2018). One may suspect that the rate of diagnostic error would be
relatively low in first-world countries. However, a relatively recent article has found
that in America, the rate of diagnostic error is 5% or 12 million Americans per year
(Singh et al., 2014). Furthermore, another American study suggested that the direct
and indirect financial cost of misdiagnosis is around $45-60 billion (Newman-Toker et
al., 2013). These findings can be extrapolated to other first-world countries such as
Canada, Australia, France, and other countries. Thus, organizations such as the
Misdiagnosis Association and Research Institution (MARI) have taken initiative to
limit the rate of misdiagnosis.
About Misdiagnosis Association and Research Institute
MARI is an organization that is dedicated to conducting research on misdiagnosis,
which is a helpful tool for healthcare providers and the general population. One of
MARI’s goals is to produce small subsections on different diseases that healthcare
providers can refer to when diagnosing their patients. This is not a primary diagnostic
tool, instead, it provides the healthcare
provider with key information about the disease to increase their ability to correctly
provide a diagnosis. Through this process, we limit the occurrence of diagnosis bias,
which is when healthcare providers provide a diagnosis without having sufficient
information (Esteban-Zubero et al., 2017). Along with reducing the rate of
misdiagnosis, these short, informative summaries can help increase the confidence
of healthcare providers who have recently started their careers, as they can increase
the accuracy of their diagnosis by confirming that the information matches the
description provided by MARI. In the future, it may be effective to integrate these
summaries within medical schools to allow future doctors to become familiar with the
platform and to give them the opportunity to develop their diagnostic skills early in
their careers.
Along with aiding the healthcare community, these disease summaries can be an
effective tool in allowing the general population to advocate for their health. For
example, the descriptions provided by MARI can educate the patient about the
different symptoms associated with different diseases. Thus, when discussing their
condition with their healthcare provider, they are able to use the appropriate jargon
and highlight key symptoms, which can guide the provider towards providing the
correct diagnosis (Judson et al., 2013). Thus, to be able to tackle the difficult
challenge of misdiagnosis, it is important to have effective collaboration between the
healthcare providers and the patient. Fortunately, the tools provided by MARI can
benefit both parties and can lead to lower rates of misdiagnosis.
Along with lowering the rates of misdiagnosis, the descriptions provided by MARI
can educate the patients about the underlying biological aspect of their condition and
to better inform them about their treatment plan. A key aspect of an effective
treatment plan is to ensure that the patient understands their medication and why
certain treatment plans are prescribed. However, a 2019 article found that 95% of
patients were not aware of the most common side effects of their
medication, which highlights the need to educate the general population on medical
conditions (Yadav et al., 2019). Once again, the description provided by MARI can
be an effective tool in filling the knowledge gap that seems to be present.
Alongside the important role that MARI plays in the healthcare community, MARI has
a vast number of different branches which focus on different topics, such as
pharmaceutical, law, nutrition, robotics, and technology, to name a few. One of
MARI’s unique and growing fields is the branch of veterinary. In many parts of the
world, including Canada, a trip to the veterinary must be paid for by the owner which
can be expensive. Having access to informative and comprehendible material allows
pet owners to familiarize themselves with common pet medical conditions. As a
result, the owner can determine which symptoms are common amongst pets and at
which point is it an appropriate time to take their pet to the vet. This can be an
effective financial saving tool for pet owners and can simultaneously reduce the wait
times and burden on the veterinary healthcare system by only treating pets that have
symptoms that may be an indication of a serious condition. In each section, MARI
has dedicated time and resources to help improve the field by providing valuable
information that can be helpful to the professionals in that field along with the general
public. It is evident that MARI has and will continue to have a positive global impact
in the vast number of fields it is involved in.
MARI is a multifaceted organization; however, one of its main contributions is in the
field of healthcare. MARI provides informative descriptions of medical conditions that
aid in reducing the rate of misdiagnosis. These summaries educate the patient about
their condition and allow them to have more effective communication with their
healthcare provider, while simultaneously allowing them to act as a health advocate.
Along with the patient, the healthcare provider also benefits from these summaries
as they can confirm that the observed symptoms